Home » About
About Europe’s leading manufacturer of custom engineered foam and thermoplastic components
AQF Medical work in some of the most demanding sectors of the medical device industry and so we understand the unique challenges our customers face in developing medical devices. Many of the foam and thermoplastic products that we fabricate become critical components within cutting edge medical devices.
With almost 40 years experience in designing and developing technical foam solutions AQF Medical have the knowledge and expertise to support your product development process.
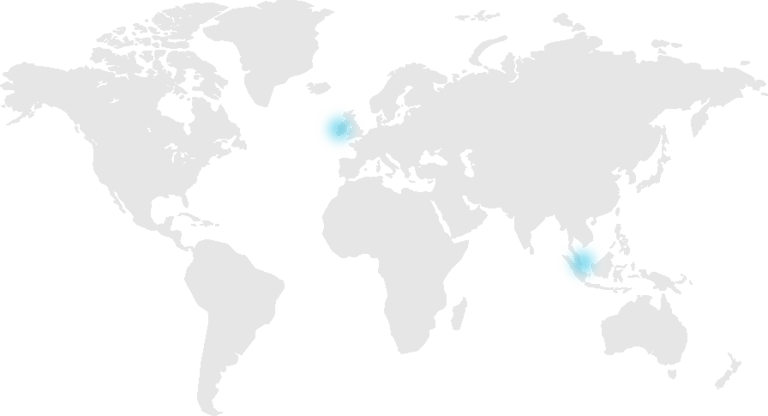
With manufacturing facilities in Ireland and Singapore we are ideally positioned to support our global customers. Our European manufacturing facility is located 50km from Dublin and our team of engineers offer unparalleled experience and expertise in foam fabrication from design through prototype stage to volume supply.

Our customer centric approach ensures we deliver the highest standards of quality and service. We strive to build and maintain strong relationships with our customers and we deliver on our promises. AQF Medical are ISO 13485 and ISO 9001 certified.
Key Market Applications
- Cross linked closed cell polyethylene packaging materials to protect Orthopaedic Implants
- Reticulated polyurethane foam components for Negative Pressure Wound Therapy
- Reticulated foams with a wide range of porosities for use in adult and paediatric Oxygenators
- Heat & Moisture Exchange foams for Anaesthesia and Intensive Care breathing devices
- Polyurethane foam with excellent absorption properties for Advanced Wound Care dressings
- Felted reticulated foams offering excellent fluid release properties for Surgical Skin Prep Perioperative Patient Care
- Soft foam pads that can help relieve the discomfort women feel during a Mammogram
- Polyethylene foams for the retention of povidone-iodine liquid used to isolate the connector of the solution transfer set for peritoneal dialysis patients

Controlled Cleanroom Environment
AQF Medical manufacture in ISO Class 8 cleanrooms which are enclosed and environmentally-controlled spaces in which temperature, humidity, pressure and contaminant levels are kept within strict limits. The controlled environment provided by our cleanrooms helps to ensure that products remain under controlled contamination levels throughout the production process.Contact Us
If you have a query relating to our company or products, or how we may be able to help you with your specific requirements, please complete the accompanying form and we will get back in touch with you.
